Translate this page into:
Surgeon-performed ultrasound analysis of the status of the vocal cords of post-thyroidectomy patients with intraoperative neuromonitoring in a tertiary referral center

*Corresponding author: Sabaretnam Mayilvaganan, Associate Professor, Department of Endocrine Surgery, Sanjay Gandhi Post Graduate Institute of Medical Sciences, Lucknow, Uttar Pradesh, India. drretnam@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Mahalakshmi VD, Mayilvaganan S, Ravisankar M, Mathialagan A, Yadav RR, Mishra P, et al. Surgeon-performed ultrasound analysis of the status of the vocal cords of post-thyroidectomy patients with intraoperative neuromonitoring in a tertiary referral center. Int J Mol Immuno Oncol 2020;5(3):121-6.
Abstract
Objectives:
Recurrent laryngeal nerve (RLN) palsy constitutes one of the dreaded complications of thyroid surgery. Sequelae of RLN injury may affect quality of life and can sometimes be life threatening. Pre-, intra-, and post- operative monitoring of the RLN is desirable for safe thyroid surgery. Surgeon-performed transcutaneous laryngeal ultrasound (TLUS) may become a substitute for laryngoscopy in the evaluation of vocal cords after thyroidectomy and may be used for prognostic correlation of intraoperative neurophysiologic monitoring (IONM). In this study, a retrospective cohort of patients who underwent thyroidectomy with monitoring of the RLN by intermittent IONM (I-IONM) was subjected to post-operative ultrasound assessment of vocal cords for the evaluation of RLN integrity.
Material and Methods:
This was a prospective study of a retrospective cohort in a tertiary care endocrine surgery unit. Fifty-six patients who underwent thyroidectomy with RLN monitoring from March 2017 to March 2019 were analyzed. Patients were analyzed during follow-up either when they arrived at the endocrine surgery outpatient department or through a telephone call. All patients underwent pre-operative laryngoscopy, intraoperative monitoring with I-IONM and Nerve Integrity Monitor 3.0 monitor, and post-operative TLUS evaluation of vocal cords.
Results:
Of the 56 patients evaluated in this study, 112 vocal cords and 102 nerves at risk were analyzed. The incidence of vocal palsy was 7.84%. The sensitivity of ultrasonography for vocal cord analysis was 85%, specificity was 95.23%, positive predictive value was 85%, and negative predictive value was 95.23% in that series. The vocal cord visualization rate was 93.5%.
Conclusion:
Surgeon-performed ultrasound may be used to assess RLN integrity in post-thyroidectomy patients to enhance post-operative care.
Keywords
Central compartment neck dissection
External branch of laryngeal nerve
Hemithyroidectomy
Intermittent Intraoperative nerve monitoring
Total thyroidectomy
Translaryngeal ultrasound
Thyroidectomy difficulty scale
Recurrent laryngeal nerve
Selective neck dissection
Vocal fold paresis
INTRODUCTION
Recurrent laryngeal nerve (RNL) palsy is one of the common complications of thyroid surgery and can affect the quality of life and can sometimes be life threatening. The incidence of temporary RLN palsy complications ranges from 3% to 12%.[1] The incidence of long-term RLN complications, such as permanent palsy, remains largely unknown as the post- operative assessment of vocal cords is not done on a routine basis. Intraoperative neuromonitoring (intraoperative neurophysiologic monitoring [IONM]) in thyroid surgery has added a functional and dynamic perspective by demonstrating that a structurally intact RLN may not necessarily correlate with functionally intact nerve.
There is no Class I evidence demonstrated with other surgical procedures using IONM, including spinal, vascular, and brain surgery.[2-5] Therefore, when using IONM, this adjunct should be recognized at the same level of evidence applied to most clinical practices pertaining to thyroid surgery.[6] IONM is rarely based on Class I evidence because it is infrequently used to prevent RLN deficits since most of the times the operating surgeon has to visualize the RLN and the incidence of permanent RLN palsy is low.[7,8] The reliability of IONM has diminished due to the incidence of false-negative and false-positive results pertaining to RLN palsy despite intraoperatively preserved, changed, or lost electromyography (EMG) signals.[9]
Pre- and post-operative laryngoscopy (L1 and L2) can be helpful for the assessment of IONM, as pre-existing RLN paresis might be associated with normal stimulation EMG. [10] The gold standard procedure for assessing vocal cord mobility is laryngostroboscopy, which is partly invasive and costly. Non-invasive ultrasonography (USG) conducted by thyroid surgeons is useful for vocal cord assessment. Transcutaneous laryngeal ultrasound (TLUS) is emerging as a potential bedside tool to map the airways during perioperative period and in critical care settings.[11] The surgeon-performed TLUS may become a substitute for invasive laryngoscopy in the evaluation of vocal cords post- thyroidectomy and may be used for prognostic correlation of IONMs. In this prospective study the endocrine surgeons performed ultrasound evaluation of Vocal cords in a cohort of patients who underwent thyroidectomy with intermittent nerve monitoring (I-IONM).
MATERIAL AND METHODS
This was a prospective study of a retrospective cohort in a tertiary care endocrine surgery unit. Fifty-six patients who underwent thyroidectomy with RLN monitoring from March 2017 to March 2019 were analyzed. All patients underwent pre-operative laryngoscopy, intraoperative neuromonitoring with I-IONM and Nerve Integrity Monitor 3.0 monitor, and post-operative ultrasound for the assessment of vocal cords. Age, gender, benign versus malignant pathology, primary versus redo surgery, weight of goiter, pre-operative vocal palsy, toxic versus non-toxic findings, and unilateral versus bilateral palsy were recorded.
Endocrine surgeons conducted post-operative vocal cord assessments by means of a TLUS using an 8–10 MHz probe. Subjects were made to lie on their backs with their neck slightly extended as their vocal cords were assessed in the axial (mid-transverse) view. Recorded videos of the ultrasounds were reviewed by a radiologist to corroborate findings.
For each patient, the probe was placed over the laryngeal prominence of the thyroid cartilage, including arytenoids in the axial view [Figure 1]. Figure 1 also shows an arrow along air vocal cord mucosa interface, seen as hyperechoic line, paretic right cord with medial displacement of vocal process of the right arytenoid. Vocal cord movement, symmetry during adduction, and abduction were observed in B-mode. Movement of vocal cords was assessed during quiet breathing (passive), phonation, and Valsalva maneuver, as applicable. Vocal fold movements in each case were video recorded and analyzed by a dedicated radiologist. The number of non-assessable, normal, and paretic cords was also recorded. Cases with decreased or asymmetric vocal cord mobility and those that could not be assessed by an ultrasound were subjected to video laryngoscopy (VDL) for further assessment by an ENT surgeon.
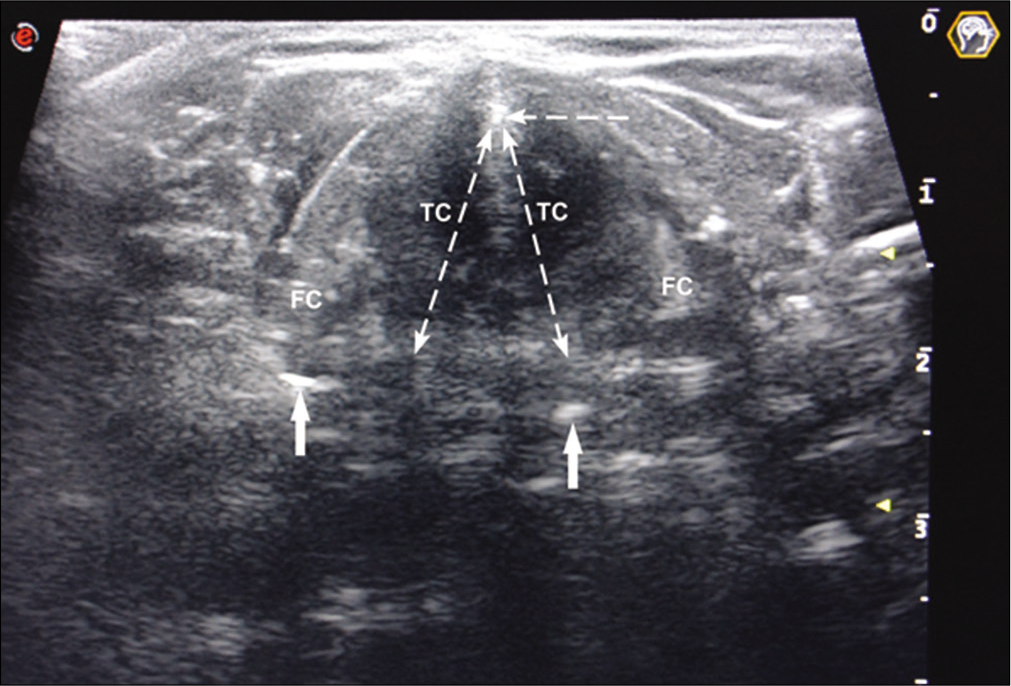
- FC – False cords, TC – true cords, solid arrows – arytenoids, horizontal broken arrow – thyroid cartilage prominence.
The statistical tests used in this study included an independent samples t-test for comparison of mean age and post-operative interval, Mann–Whitney U-test for the median weight of goiter, Cohen’s kappa to assess for within- group variability, Chi-square tests, and Fisher’s exact tests for comparison of various demographic and clinical variables between groups with normal and abnormal vocal cord movements on translaryngeal USG following thyroidectomy.
RESULTS
Fifty-six patients with 112 vocal cords and 102 nerves at risk underwent TLUS evaluation of vocal cords post-thyroidectomy. As can be deduced from the demographic data represented in Table 1, the gender ratio of the patients in this study was 4.6:1 and the mean age was around 41 years. Descriptive data outlined in Table 1 indicate that most of the cases were benign. A majority of the cases in this study were reportedly euthyroid, while the remainder mostly consisted of hyperthyroid cases and relatively few cases of subclinical hyperthyroidism. Majority of these patients underwent total thyroidectomy (TT), while others underwent hemi-thyroidectomy (HT), TT with central compartment neck dissection and selective neck dissection (TT + CCLND + SLND), and TT + CCLND (of descending frequency). Most cases involved primary surgeries, with two cases of repeat procedures. The median weight of goiter in the series was 67.25 g. Vocal cord status was analyzed post-thyroidectomy in all patients by surgeon- performed TLUS during follow-up visits. All patients underwent thyroidectomy with IONM and were assessed as having considerably difficult thyroidectomies on the modified thyroidectomy difficulty scale.[12] The vocal cord visualization rate was 93.5%. As shown in Table 2, four cases were non-assessable by ultrasound in this series.
| Variable’s | VC palsy (n=8) (%) | BLVC mobile (n=48) (%) | Total (n=56) (%) | P value |
|---|---|---|---|---|
| Number of males | 2 (25) | 8 (16.7) | 10 (17.9) | 0.623 |
| Number of females | 6 (75) | 40 (83.3) | 46 (82.1) | 0.623 |
| Malignant | 4 (50) | 9 (18.8) | 13 (23.2) | 0.074 |
| Benign | 4 (50) | 39 (81.2) | 43 (76.8) | 0.074 |
| Mean age (years) £ | 34.50±10.994 | 41.96±13.278 | 40.89±13.152 | 0.139 |
| Toxic | 1 (12.5) | 11 (22.9) | 12 (21.4) | 0.756 |
| Subclinical hyperthyroidism | 0 | 2 (4.2) | 2 (3.6) | 0.756 |
| Non-toxic | 7 (87.5) | 35 (72.9) | 42 (75) | 0.756 |
| HT | 0 | 10 (21.3) | 10 (18.2) | 0.020 |
| TT | 3 (37.5) | 30 (63.8) | 33 (60) | |
| TT+CCNLD | 1 (12.5) | 2 (4.3) | 3 (5.5) | |
| TT+CCLND+SLND | 4 (50) | 5 (10.6) | 9 (16.4) | |
| Primary surgery | 8 (100) | 46 (95.8) | 54 (96.4) | 0.732 |
| Pre-operative VC Palsy | 1 (12.5) | 1 (2.1) | 2 (3.6) | 0.051 |
| Pre-operative phonatory gap | 1 (12.5) | 0 | 1 (1.8) | |
| Median weight of goiter (g) ¥ | 48.5 (17, 76.8) | 86.5 (20, 218) | 67.25 (19.75, 199.25) | 0.199 |
£-Independent sample t-test, ¥- Mann–Whitney U-test, Chi-square/Fisher’s exact test for all other variables. P<0.05. TT: Total thyroidectomy, HT: Hemithyroidectomy, CCLND: Central compartment neck dissection, SLND: Selective neck dissection
| Age/sex | Diagnosis/surgery | USG view non-assessable – transverse view | VDL |
|---|---|---|---|
| 48/M | Graves/TT | Difficulty due to angulation of thyroid cartilage | Rt. VC paresis Normal Lt VC |
| 60/M | MNG/TT | Angulated cartilage with difficulty in probe placement | B/L VC mobile |
| 50/M | MNG /TT | Calcified cartilage | B/L VC mobile |
| 56/F | Toxic MNG with tracheomalacia/TT+Tracheostomy | Lt VC not well visualized Partly calcified cartilage? post-tracheostomy changes? paretic | B/L VC mobile |
USG: Ultrasonography, TT: Total thyroidectomy, VDL: Video laryngoscopy
Eight unilateral vocal cord palsy cases diagnosed by translaryngeal ultrasound were subjected to VDL. Among these, six cases were confirmed to have unilateral palsy, one case diagnosed to have unilateral palsy was found to have U/L palsy with paresis on other side, and the another case was found to have normal mobility on VDL. Two patients had pre-operative vocal palsy, with one case demonstrating improvement postoperatively with a benign etiology, and the another case demonstrating persistent palsy with a malignant etiology. Cohen’s kappa was used to assess variability within the group of 14 patients who underwent both USG and VDL. Results showed substantial agreement between USG and VDL with a kappa value of 0.721 (P = 0.002).
DISCUSSION
The series of difficult thyroidectomy presented in this study was found to have 7.8% incidence of post-operative vocal palsy. This is similar to the results of a meta-analysis indicating a post-surgery VCP rate of 9.8% among more than 25,000 patients[13] and a unilateral VCP rate of 9% among 5000 Medicare beneficiaries who underwent thyroidectomy.[14]
RLN palsy correlates with the extent of the performed surgery, with the highest incidence noted in patients who underwent TT with CCLND and SLND. In a series by Chen et al.,[15] the incidence of unilateral vocal fold paralysis (UVFP) and involvement of the external branch of the superior laryngeal nerve (EBSLN) differed among surgery types and were highest among patients undergoing TT with neck dissection (TTND). TTND is associated with higher risks of UVFP and EBSLN injury, 6.3% and 3.2%, respectively, than other types of surgery, implying the need for intraoperative nerve monitoring in these high-risk situations.
Vocal cord visualization rate using surgeon-performed ultrasound was 93.75% compared with 96.9% in a study by Kumar et al.[16] With the middle transverse procedure, the visualization was 70.2% and with the lateral vertical procedure, the visualization of arytenoid cartilage was 98.4% in a study by Fukuhara et al.[17] About 6.25% vocal cords were non-assessable by transcutaneous laryngeal USG on transverse view which may necessitate special complex maneuvers such as utilization of water bath and longitudinal views. Seven out of eight patients’ cords identified by USG were proven to be paretic by VDL.
Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of SUS for vocal cord analysis were 85%, 95.23%, 85%, and 95.23%, respectively, in this series. Other reported literature for SUS sensitivity, specificity, PPV, and NPV being 93.3%, 97.8%, 77.8%, and 99.4% by Wong et al.[18] in 204 patients; 33%, 95%, 42%, and 89% by Borel, Delemazure, Espitalier, Spiers, and Mirallie in 2016;[19] and 83.33%, 97.9%, 76.92%, and 98.59% by Sadacharan, Ferdinant, Rakeshchandru, and Thalavai [Table 3].[20]
| Study | Sensitivity (%) | Specificity (%) | PPV (%) | NPV(%) |
|---|---|---|---|---|
| Wong et al.[18] | 93.3 | 97.8 | 77.8 | 99.4 |
| Borel et al.[19] | 33 | 95 | 42 | 89 |
| Sadacharan et al.[20] | 83.33 | 97.9 | 76.92 | 98.59 |
| Present study | 85 | 95.23 | 85 | 95.23 |
PPV: Positive predictive value, NPV: Negative predictive value
Peculiar irregular wriggling movements of the vocal cords were identified in few of our cases, mostly in cases with paretic cords during the recovery phase. Reverberation artifacts at the air mucosa interface accentuated by turbulent laryngeal airflow or ineffective movements due to synkinesis in vocal palsy may be responsible for this phenomenon. Dysphonia in UVFP is characterized by breathiness or the perception of turbulent laryngeal airflow.[21] This air leak leads to a weak voice and increased phonatory effort for the patient. The vibrational asymmetry caused by UVFP leads to the appearance of subharmonics and the perception of diplophonia or roughness. One hypothesis in the physiopathology of recurrent nerve paralysis is based on the phenomenon of synkinesis,[22] in which axons are rerouted during their growth after injury in such a way that axons initially innervating adductor muscles end up innervating the abductor muscle and vice versa. The resulting vector of the mobility of the vocal fold is zero because the antagonistic muscles contract at the same time. Thus, small movements of the paralyzed vocal fold may be observed without an effective voice. The interarytenoid muscle continues to function due to its bilateral innervation. The paralyzed larynx may spontaneously reinnervate from the contralateral side through endolaryngeal neural anastomoses.[23] Reoccupation of vacant motor endplates by autonomic nerves has also been shown to occur.[24] Vocal muscle atrophy with periarticular fibrosis causes lateral retraction of the paralyzed vocal fold. Other laryngeal structures, such as the ventricular folds and the epiglottis, may show significant compensatory motion during phonation.[25] Finally, even laryngeal spasms with consequences on voice and breathing have been shown to occur after URLNP.[26]
In a situation of difficult thyroidectomy with extensive dissection in the central compartment where RLN is at a very high risk of neuropraxia, the surgeon should have all options available in the armamentarium to have a safe outcome, including IONM. Post-operative USG can be a safe and non-invasive option for the surgeon to evaluate the vocal cords and closely follow-up in the event that any palsy is discovered. Ultrasound is widely available and cheap compared to VDL. We also found that post-thyroidectomy laryngeal changes and position of tracheal shift can hinder vocal cord visualization.
The strength of our study includes the fact that the vocal cord status has been monitored preoperatively by laryngoscopy, intraoperatively by I-IONM, and postoperatively by ultrasound to determine RLN integrity in all these cases, thereby preventing unnecessary invasive procedures. Our study had some limitations. The sample size is adequate, but a large sample size can provide many new insights. Not all cases analyzed by TLUS underwent VDL, which is an invasive procedure performed by ENT surgeons and patients had to take prior appointment. In our department routine, post-operative VDL is not recommended since it is invasive and associated with extra cost. Early recovery of VC function and non-permanent palsy was expected in TLUS missed vocal palsy cases.[27] Various other approaches, such as the lateral approach, described by Fukuhara et al.,[17] evaluation of vocal fold displacement velocity as described by Dedecjus,[28] and usage of gel pad[29] could have been utilized, especially in male patients or patients with ossification of the thyroid cartilage and in non-assessable cases other than the transverse view. This may be considered when conducting similar research in future.
CONCLUSION
Surgeon-performed ultrasound can be a useful tool in the post-operative setting to analyze the vocal cords and to benefit patients who have palsy by means of recommending close follow-ups and early intervention if necessary. Since ultrasound has become a part of clinical examination and also widely available it can also used for post operative assessment of vocal cords.
Patient consent declaration
Institutional Review Board (IRB) permission obtained for the study.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Epidemiology of vocal fold paralyses after total thyroidectomy for well-differentiated thyroid cancer in a Medicare population. Otolaryngol Head Neck Surg. 2014;150:548-57.
- [CrossRef] [PubMed] [Google Scholar]
- Intraoperative neurophysiology is here to stay. Childs Nerv Syst. 2010;26:413-7.
- [CrossRef] [PubMed] [Google Scholar]
- Timing of surgery for aneurysmal subarachnoid haemorrhage. Cochrane Database Syst Rev. 2001;2:CD001697.
- [CrossRef] [PubMed] [Google Scholar]
- Review of treatment trials in human spinal cord injury: Issues, difficulties, and recommendations. Neurosurgery. 2006;59:957-82.
- [CrossRef] [PubMed] [Google Scholar]
- Cost-effectiveness of intraoperative facial nerve monitoring in middle ear or mastoid surgery. Laryngoscope. 2003;113:1736-45.
- [CrossRef] [PubMed] [Google Scholar]
- Intraoperative neural monitoring in thyroid surgery: Role and responsibility of surgeon. J Endocr Surg. 2018;18:49.
- [CrossRef] [Google Scholar]
- Continuous intraoperative neural monitoring in thyroid surgery: A Mexican experience. Updates Surg. 2017;69:505-8.
- [CrossRef] [PubMed] [Google Scholar]
- Intraoperative monitoring of the recurrent laryngeal nerve in thyroid surgery. World J Surg. 2008;32:1358-366.
- [CrossRef] [PubMed] [Google Scholar]
- Prospective study on loss of signal on the first side during neuromonitoring of the recurrent laryngeal nerve in total thyroidectomy. Br J Surg. 2013;100:662-6.
- [CrossRef] [PubMed] [Google Scholar]
- Postoperative laryngoscopy in thyroid surgery: Proper timing to detect recurrent laryngeal nerve injury. Langenbecks Arch Surg. 2010;395:327-31.
- [CrossRef] [PubMed] [Google Scholar]
- Ultrasound: A promising tool for contemporary airway management. World J Clin Cases. 2013;3:926-9.
- [CrossRef] [PubMed] [Google Scholar]
- Patient, thyroid, and surgeon related factors that make thyroidectomy difficult-cohort study. Ann Med Surg (Lond). 2019;49:14-18.
- [CrossRef] [PubMed] [Google Scholar]
- Diagnosis of recurrent laryngeal nerve palsy after thyroidectomy: A systematic review. Int J Clin Pract. 2009;63:624-9.
- [CrossRef] [PubMed] [Google Scholar]
- Nationwide variation in rates of thyroidectomy among US Medicare beneficiaries. JAMA Otolaryngol Head Neck Surg. 2017;143:1122-5.
- [CrossRef] [PubMed] [Google Scholar]
- Risk factors for thyroid surgery-related unilateral vocal fold paralysis. Laryngoscope. 2019;129:275-83.
- [CrossRef] [PubMed] [Google Scholar]
- Assessment of functionality of vocal cords using ultrasound before and after thyroid surgery: An observational study. Indian J Anaesth. 2018;62:599-602.
- [CrossRef] [PubMed] [Google Scholar]
- A novel lateral approach to the assessment of vocal cord movement by ultrasonography. World J Surg. 2018;42:130-6.
- [CrossRef] [Google Scholar]
- A prospective, assessor blind evaluation of surgeon-performed transcutaneous laryngeal ultrasonography in vocal cord examination before and after thyroidectomy. Surgery. 2013;154:1158-65.
- [CrossRef] [PubMed] [Google Scholar]
- Transcutaneous ultrasonography in early postoperative diagnosis of vocal cord palsy after total thyroidectomy. World J Surg. 2016;40:665-71.
- [CrossRef] [PubMed] [Google Scholar]
- The utility of surgeon performed transcutaneous laryngeal ultrasound for the assessment of vocal cords in post-thyroidectomy patients. Int Surg J. 2018;5:3505-9.
- [CrossRef] [Google Scholar]
- Objective acoustic and aerodynamic measures of breathiness in paralytic dysphonia. Eur Arch Otorhinolaryngol. 2003;260:175-82.
- [CrossRef] [PubMed] [Google Scholar]
- The innervation of the human larynx. Arch Otolaryngol Head Neck Surg. 1993;119:934-9.
- [CrossRef] [PubMed] [Google Scholar]
- Synapse formation by autonomic nerves in the previously denervated neuromuscular junctions of the feline intrinsic laryngeal muscles. Brain Res. 1991;539:276-86.
- [CrossRef] [Google Scholar]
- Ventricular dysphonia: A profile of 40 cases. Laryngoscope. 1992;102:1296-301.
- [CrossRef] [PubMed] [Google Scholar]
- Paroxysmal laryngospasm after laryngeal nerve injury. Laryngoscope. 1999;109:694-7.
- [CrossRef] [PubMed] [Google Scholar]
- Laryngeal synkinesis: Its significance to the laryngologist. Ann Otol Rhinol Laryngol. 1989;98:87-92.
- [CrossRef] [PubMed] [Google Scholar]
- Vocal cord palsies missed by transcutaneous laryngeal ultrasound (TLUSG): Do they experience worse outcomes? World J of Surg. 2018;43:824-30.
- [CrossRef] [PubMed] [Google Scholar]
- Real-time, high-resolution ultrasonography of the vocal folds: A prospective pilot study in patients before and after thyroidectomy. Langenbecks Arch Surg. 2010;395:859-64.
- [CrossRef] [PubMed] [Google Scholar]
- A novel gel pad laryngeal ultrasound for vocal cord evaluation. Thyroid. 2017;27:553-7.
- [CrossRef] [PubMed] [Google Scholar]






